Proceedings Home | W2W Home 
Purpose
Animal manure contains nutrients and organic matter that are valuable to crop production. Applying manure to nearby fields can be a significant source of environmental contamination, however, if managed incorrectly. In many cases, concentrated animal production facilities are not close enough to sufficient cropland to fully utilize these resources and management of manure becomes more of a disposal issue rather than a utilization opportunity. One potential solution is to remove and concentrate manure nutrients so they can be cost effectively transported longer distances to cropland that is lacking in nutrients. The objective of this work was to design and test a pilot-scale system to implement a hydrophobic, gas-permeable, ePTFE (a synthetic fluoropolymer) membrane (U.S. patent held by USDA) to recover ammonia from swine wastewater in a solution of sulfuric acid. The pilot-scale system was designed to replicate the laboratory results and to determine critical operational controls that will assist in design of farm-scale systems.
What did we do?
Through a series of preliminary experiments, we established operational criteria and selected a membrane with an inside diameter of 0.16 in., wall thickness of 0.023 in., and a density of 0.016 lb in-3. A test system was developed (Figure 1) with 19 membrane tubes within a 2.01-inch diameter, 24.7-inch-long reactor, giving a membrane density of 3.83 sq. in. per cubic inch of reactor volume. Wastewater first passed through a CO2 stripping column (4.016 in. diameter, 55 in. length) where a small air stream (0.0614 cfm) stripped CO2 from the wastewater and raised the pH one full unit, shifting the equilibrium to NH3 and enhancing transport across the membrane. Batch tests (0.706 ft3) were run for 9-12 days with wastewater recirculating at a rate of 0.16 gpm. The recovery fluid inside the tubular membranes was a 0.01 N sulfuric acid solution with the pH automatically maintained below 4.0 standard units and recirculating at a rate of 1/100th the wastewater flow rate. Freshly collected settled wastewater and anaerobic digester effluent were tested to determine the mass of ammonia collected, the acid required to maintain the low pH of the recovery solution, and potential ammonia losses to the atmosphere.
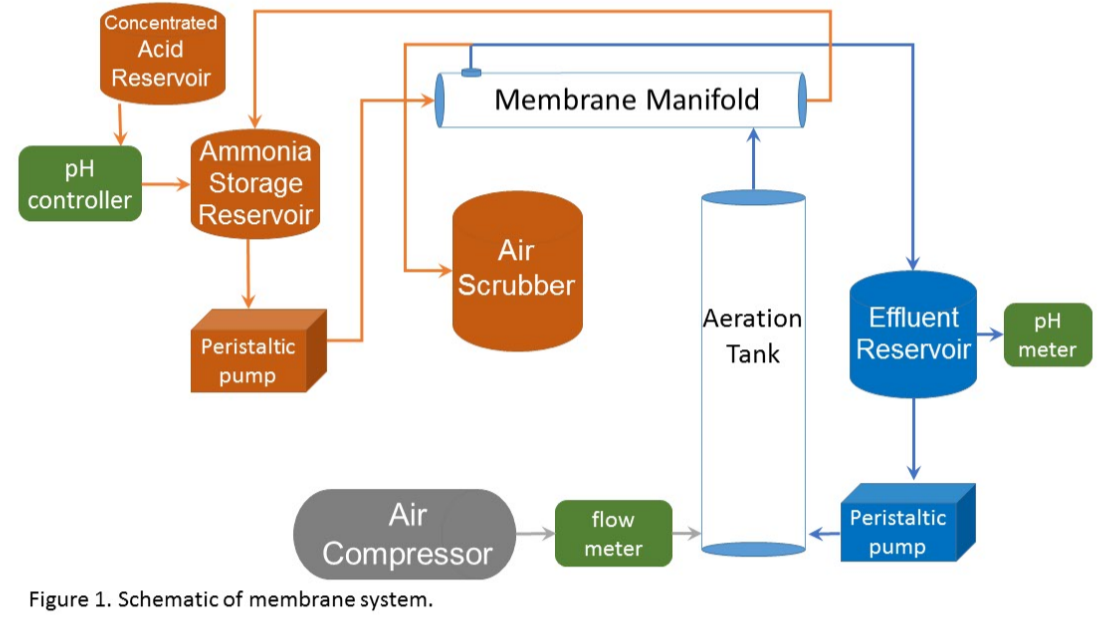
What have we learned?
The freshly collected wastewater had an initial mass of 35.6 g nitrogen but the NH3 was only 14.5 g, leading to a recovery of 11.8 g (33% of initial content) over 12 days. The anaerobic digester effluent had an initial mass of 33.2 g nitrogen with an NH3 mass of 31.3 g. The higher fraction of ammonia helped push the recovery to 25.7 g or 77% of the initial nitrogen content (see Figure 2). Very little ammonia was lost with the exhaust air.

Future Plans
An optimized membrane reactor could be a viable tool in ammonia nitrogen recovery from a manure treatment system if used in conjunction with digestion. Higher economic value could be generated by further concentrating the ammonium sulfate product.
Corresponding author, title, and affiliation
John J. Classen, Associate Professor, Biological & Agricultural Engineering, NCSU
Corresponding author email
Other authors
J. Mark Rice, Extension Specialist, NCSU; Alison Deviney, Graduate Research Assistant, NCSU
Additional information
John J. Classen
Biological and Agricultural Engineering
Campus Box 7625
North Carolina State University
919-515-6755
Acknowledgements
This project was supported by NRCS CIG Award 69-3A75-12-183. The authors are grateful for the analytical work of the BAE Environmental Analysis Laboratory, Dr. Cong Tu, manager.
